Home » Course Layouts » Free Course Layout Udemy
This subject deals primarily with equilibrium properties of macroscopic and microscopic systems, basic thermodynamics, chemical equilibrium of reactions in gas and solution phase, and macromolecular interactions.
0
English
English [CC]
FREE
- Learn basic syntax that can apply to any language.
- Learn what is a programming language and the basic concepts for beginners.
- Understand what is Javascript in it's truest form.
- Know the basic syntax of Javascript.
- Know some hidden quirks in Javascript.
Description
Course content
- Introduction to Thermo; 0th Law; Temperature; Work; Heat Unlimited
- State Functions, 1st Law, Paths Unlimited
- Joule and Joule-Thompson; Heat Capacity Unlimited
- Reversible and Irreversible Processes Unlimited
- Thermochemistry Unlimited
- 2nd Law; Entropy (Boltzmann and Clausius) Unlimited
- ΔS for Reversible and Irreversible Processes Unlimited
- Equilibrium; Maxwell Relations; Free Energy Unlimited
- Chemical Potential; Phase Equilibrium Unlimited
- Chemical Equilibrium; Equilibrium Constant Unlimited
- Standard States; Gibbs-Duhem Unlimited
- ΔG0= -RTlnK; Example Unlimited
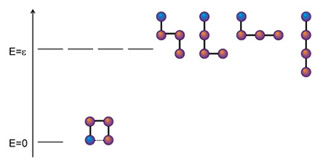
- Boltzmann Distribution Unlimited
- Thermo and Boltzmann Distribution Unlimited
- Occupation of States Unlimited
- Third Law Unlimited
- Phase Equilibria, Single Component Unlimited
- Phase Equilibria II; Clausius Clapeyron Unlimited
- Solvation; Colligative Properties Unlimited
- Osmotic Pressure and Phase Partitioning Unlimited
- Surface Tension Unlimited
- Polymer 1 – Freely Jointed Chain Unlimited
- Polymer 2 – Chain Conformation Unlimited
- Polymer 3 – Rubber Elasticity Unlimited
- Electrolyte Solutions Unlimited
- Electrolytes at Interfaces; Debye Length Unlimited
- Titration of Polyelectrolytes Unlimited
- Thermodynamics of DNA Hybridization Unlimited
- Cooperativity Unlimited
N.A
- 5 stars0
- 4 stars0
- 3 stars0
- 2 stars0
- 1 stars0
No Reviews found for this course.
Instructor
Massachusetts Institute of Technology
5
5
1
1925
1520
Explore Free Courses
Access valuable knowledge without any cost.
{"title":"","show_title":"0","post_type":"course","taxonomy":"course-cat","term":"engineering-skills,health-and-safety","post_ids":"","course_style":"free","featured_style":"course6","masonry":"","grid_columns":"clear4 col-md-3","column_width":"268","gutter":"30","grid_number":"4","infinite":"","pagination":"","grid_excerpt_length":"20","grid_link":"1","grid_search":"0","course_type":"","css_class":"","container_css":"","custom_css":""}